- Date :01 Apr, 2026
- Category : Organic Chemistry
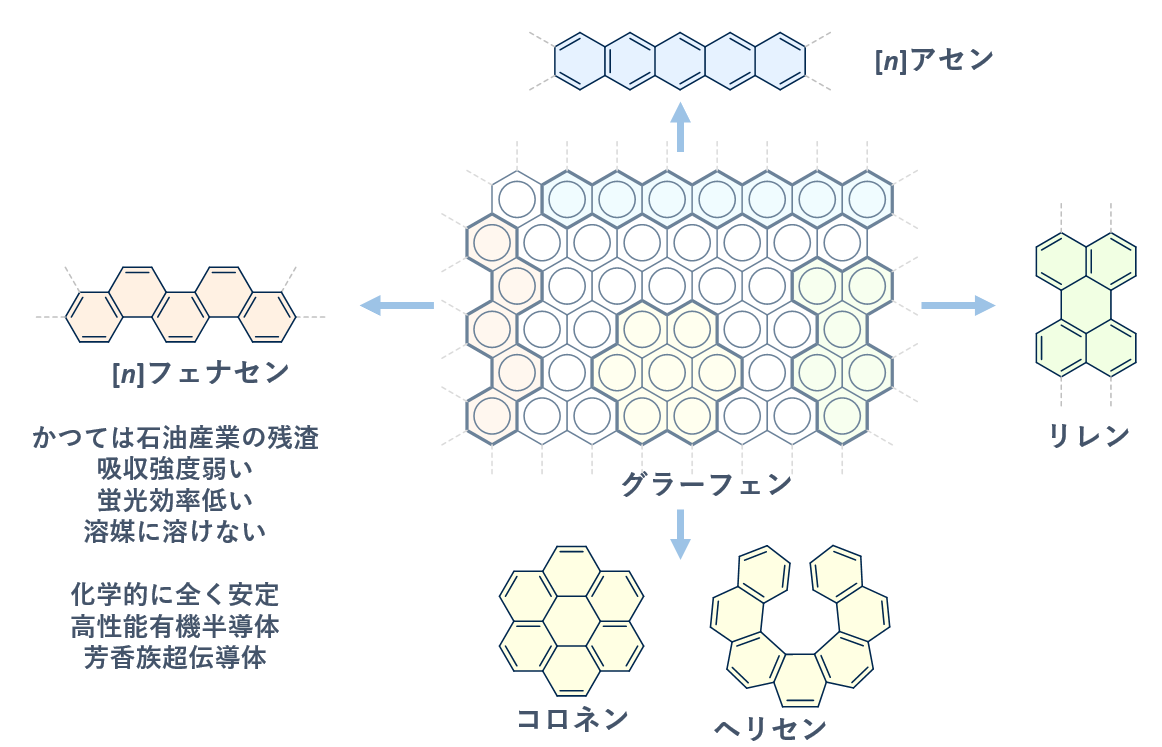
Benzene is the prototypical framework of aromatic compounds. By fusing benzene rings in various directions, we can create a wide variety of polycyclic aromatic hydrocarbons. Representative examples are shown in the figure below. When hexagonal benzene units are closely packed in a plane, they form graphene. Extracting segments from the graphene structure reveals many polycyclic aromatic hydrocarbons with diverse structural features. (Of course, these molecules are not literally cut out from graphene; instead, each is synthesized by routes tailored to its structure.) Among such molecules, [n]acenes have long attracted interest for their chemical and physical properties, and have been extensively studied (here, n denotes the number of benzene rings in the molecule). Rylenes and cyclic coronene derivatives have also been investigated as functional materials.
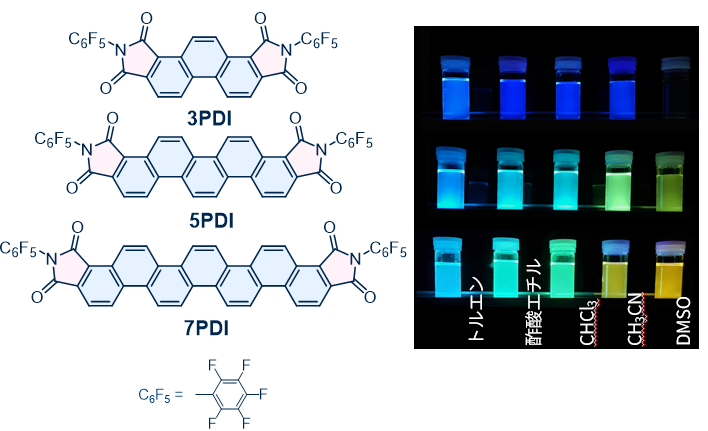
[n]Phenacenes have a zigzag array of n benzene rings and are constitutional isomers of [n]acenes. [n]Phenacenes were already known more than 100 years ago to be present in coal tar and pitch—residual materials from the petroleum industry—but they have rarely been used as functional materials. This is mainly because their absorption and fluorescence intensities are weak and their solubility is extremely low. On the other hand, [n]phenacenes are known to be chemically very robust. If we can impart functionality to such chemically durable phenacenes, it should be possible to obtain functional molecules with excellent durability and weather resistance. Our research group has previously demonstrated, ahead of the rest of the world, that [n]phenacenes can serve as active layers in organic field-effect transistors and as aromatic superconductors. More recently, we have begun exploring the use of [n]phenacenes—which originally exhibit weak fluorescence and little tunability of emission color—as functional fluorescent molecules. Globally, there have been very few studies that utilize phenacenes with n ≥ 5 as functional fluorescent dyes. For [n]phenacenes without substituents, changing the number of benzene rings from 4 to 7 yields only weak fluorescence in the near-UV to violet region. We therefore designed [n]phenacenes bearing electron-withdrawing imide groups and investigated their fluorescence properties. 3PDI, 5PDI, and 7PDI have imide units at both ends of the [n]phenacene core. Their fluorescence quantum yields increased up to about 30%, meaning that the introduction of imide groups significantly enhances fluorescence intensity compared to the parent phenacenes without imides. Furthermore, as shown in the figure below, the emission color changes visibly to the naked eye depending on the polarity of the solvent. In particular, 5PDI and 7PDI exhibit multicolor emission—from blue in toluene to yellow–orange in dimethyl sulfoxide (DMSO). These results show that by chemically modifying the [n]phenacene framework, one can construct fluorescent dyes whose emission colors respond to their environment.
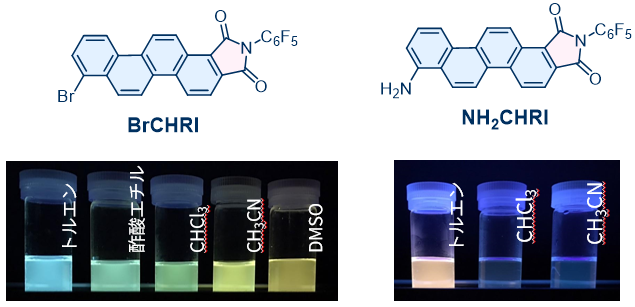
We also observed solvent-dependent changes in fluorescence color for BrCHRI and NH2CHRI, [4]phenacene (chrysene) derivatives bearing bromo and imide groups as electron-donating substituents. Notably, NH2CHRI exhibits red emission in nonpolar toluene, which we consider to be the first example of a red-emissive luminophore based on the chrysene skeleton.

The key to the solvatochromic fluorescence of [n]phenacene derivatives with imide groups lies in the polarization of the molecule in its excited state. When the degree of molecular polarization differs greatly between the ground state (before light absorption) and the excited state that emits fluorescence, properties such as fluorescence become highly sensitive to the surrounding solvent environment. Molecules of the [n]phenacene family were once regarded as rather disappointing: “insoluble, non-emissive, and with non-tunable emission colors.” Our work shows that by appropriate chemical modification, these molecules can be transformed into functional luminophores. We have also found that the chemical reactivity of [n]phenacenes themselves can be tuned. By designing new molecules, we can control the physical and chemical properties of [n]phenacenes and related polycyclic aromatic hydrocarbons, thereby contributing to the future design and engineering of multifunctional molecular materials.
For further details, please refer to:
- https://pubs.rsc.org/en/content/articlepdf/2023/ra/d2ra07771j
- https://www.sciencedirect.com/science/article/pii/S1010603024001576?via%3Dihub
- https://academic.oup.com/chemlett/article/54/5/upaf091/8128644
Want to know more about Prof. Okamoto’s research!→Synthetic and Physical Organic Chemistry Laboratory